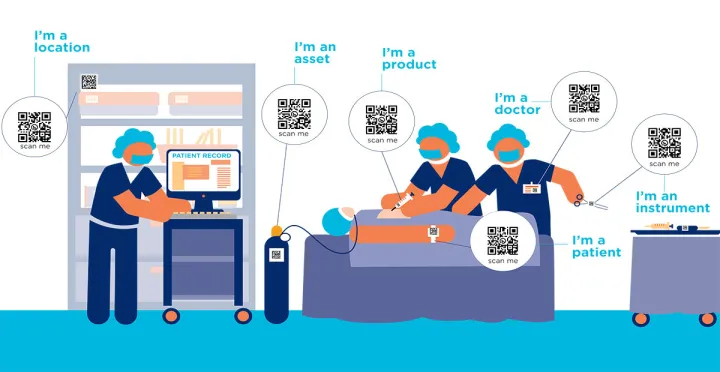
Healthcare
Globally consistent, standardised imaging, saving and analysing methods promote the merging of data streams. Real-time information produced in a common language and stored in the correct place and the compatibility of systems promote communication between occupational groups as well as up-to-date decision-making processes. This enables comprehensive cost optimisation per patient and unit, among other things.
Benefits for the healthcare industry
- The serially numbered barcode enables the drug to be automatically recorded and linked to the patient's data, as well as to ensure authenticity.
- The unique device identifier (UDI) makes the medical device a globally identifiable and traceable unit.
- The code can be used both in logistics and in clinical nursing work.
- The device can be connected to the patient's data.
- Quick recalls of medicines and supplies are possible.
Get your official UDI identifiers from GS1
In healthcare, the UDI identifier is a code placed on a device and/or packaging label. The manufacturer is expected to follow the official issuing entity’s instructions when generating and administering the identifiers. An individual identifier is required to ensure that each device can be identified and that the identifier can be used similarly across all markets.GS1 is a UDI identification organization authorized by both the European Commission and the US FDA.